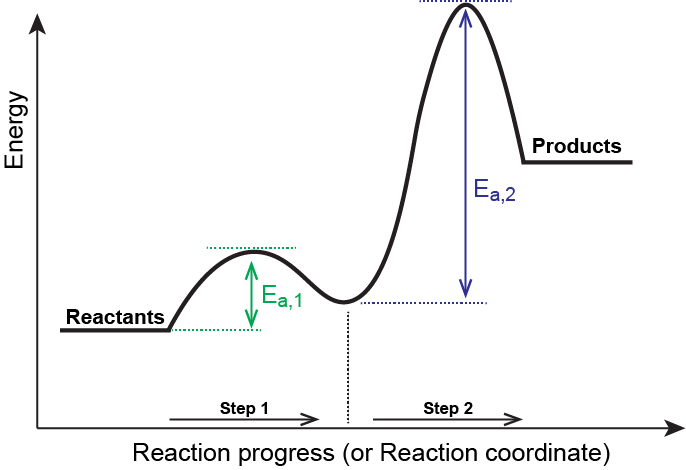
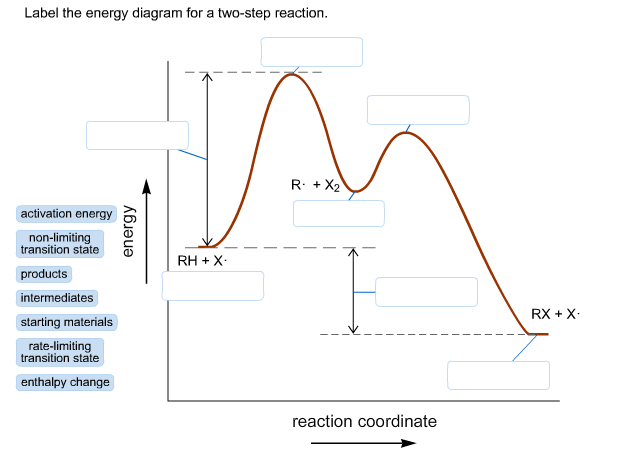
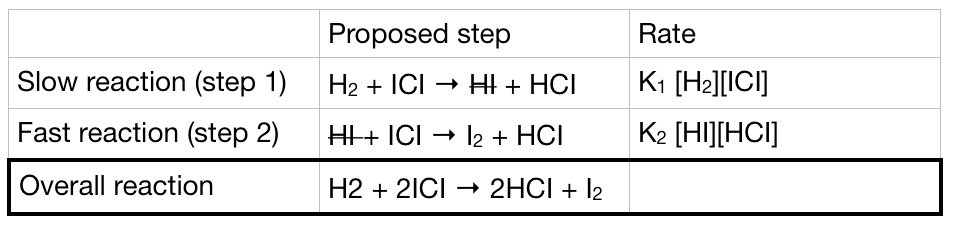
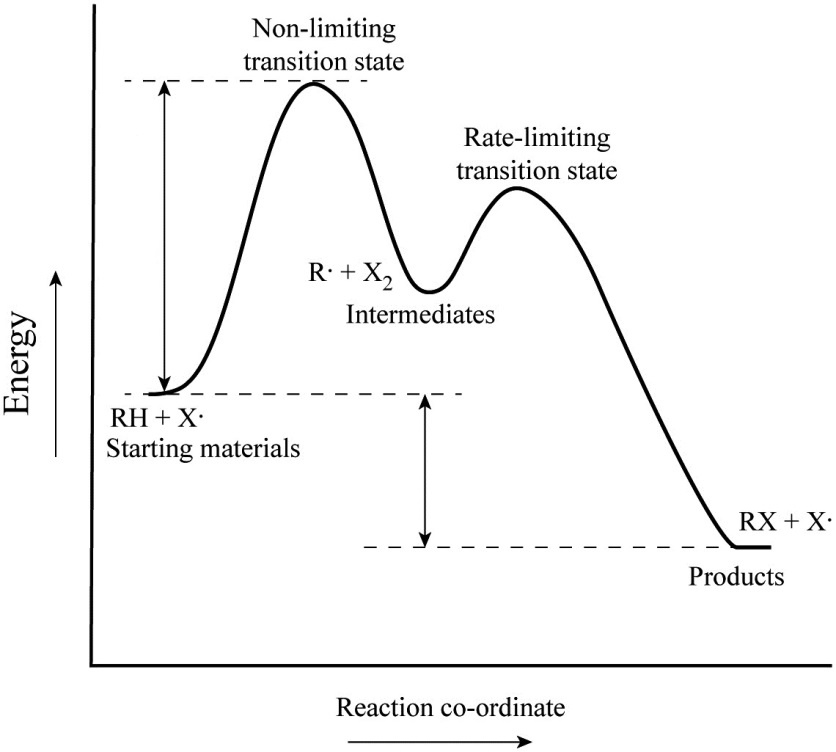
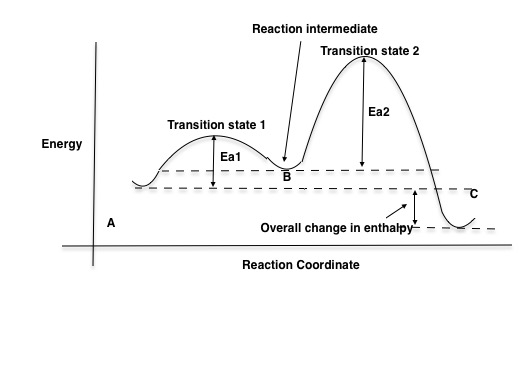
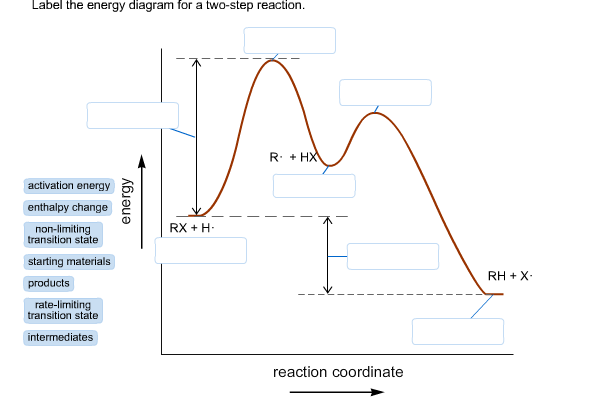
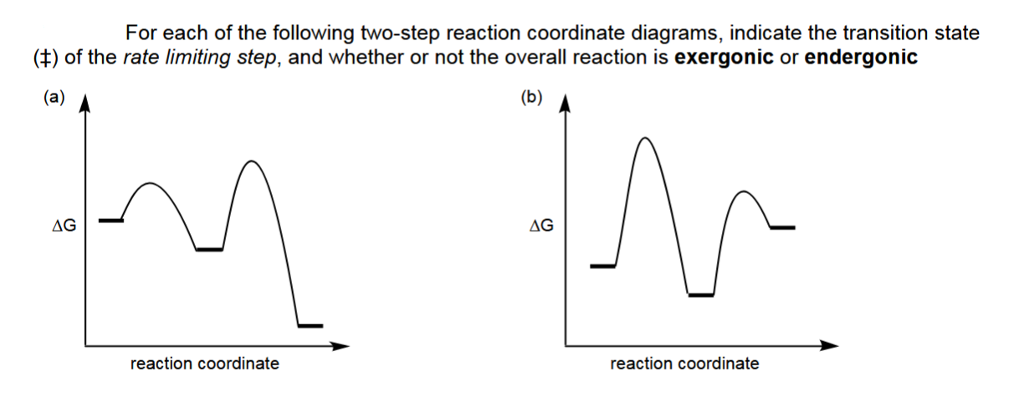
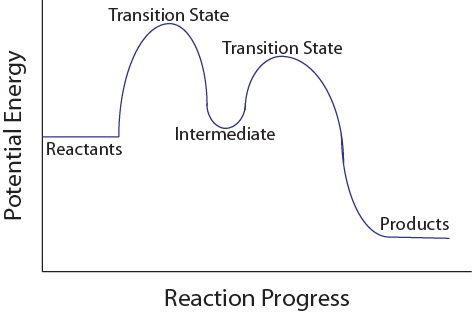
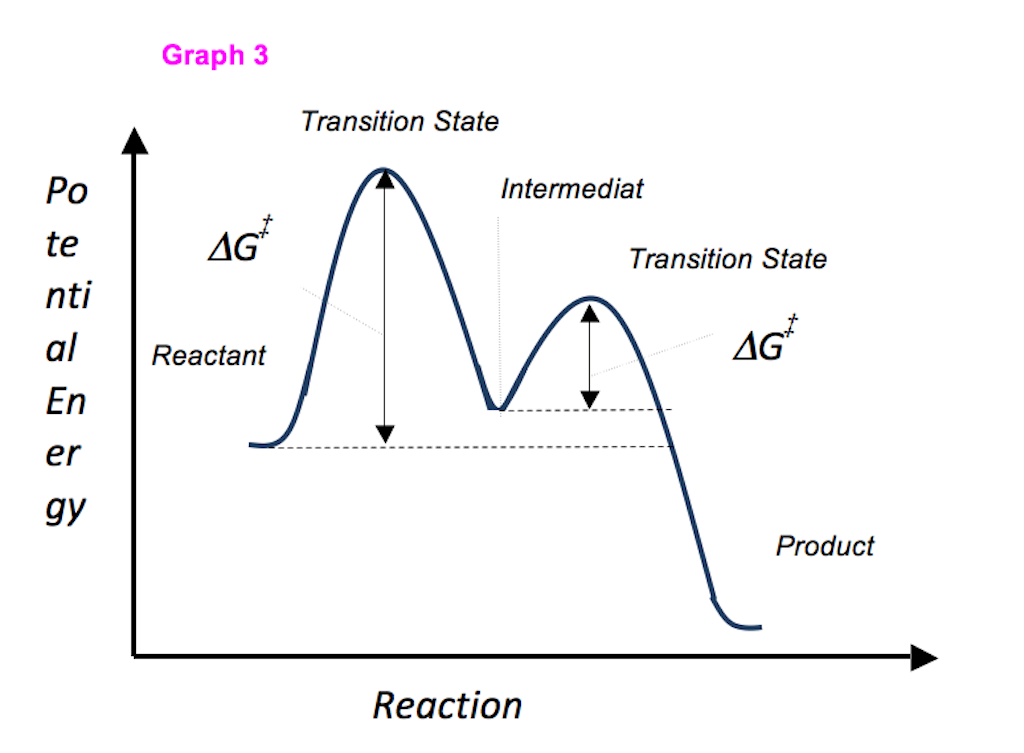
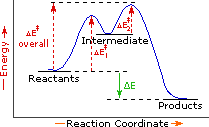
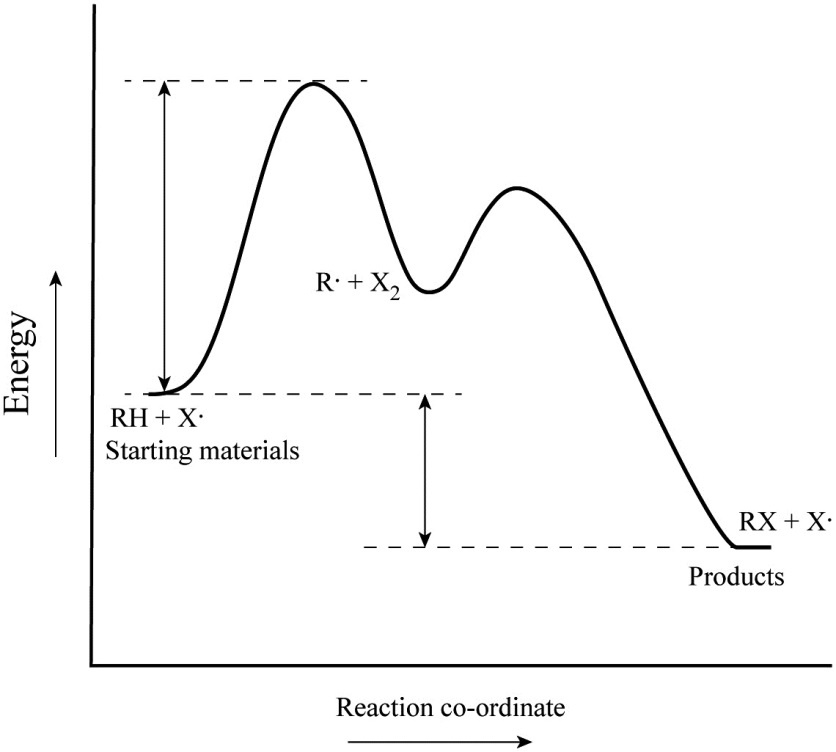
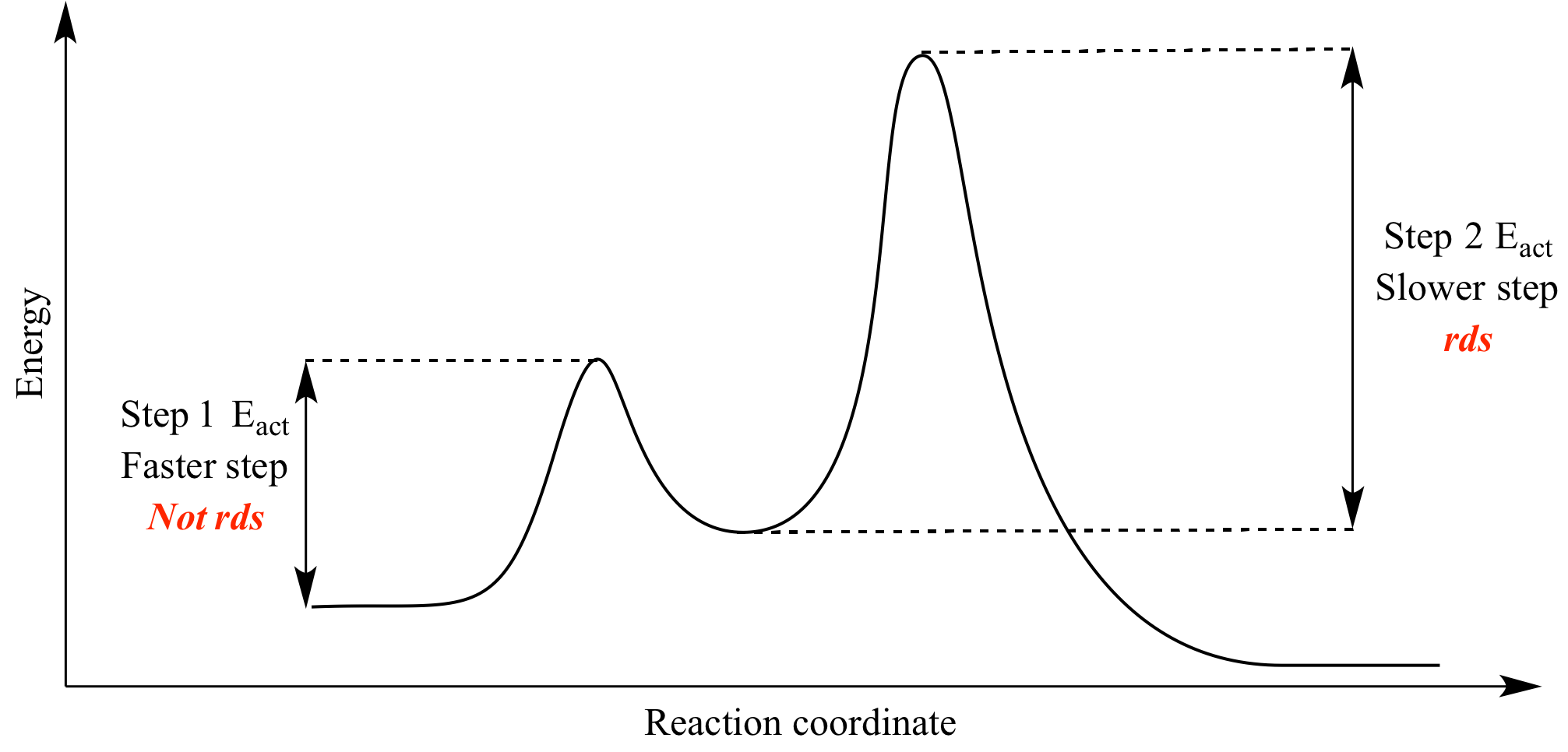
Is the activation energy for the overall reaction equal to the activation energy for the rate determining step? - Quora

Question Video: Identifying Steps in a Reaction Profile Diagram for a Two- Step Chemical Reaction | Nagwa
Many reactions proceed through a series of steps. What are the individual steps of the reaction called and on what do they depend? - Quora

Difference Between Elementary Step and Rate Determining Step | Compare the Difference Between Similar Terms
Draw an energy diagram for a two-step exergonic reaction whose second step is faster than its first step. | Homework.Study.com

SOLVED: A two-step reaction mechanism is proposed for reaction, as represented below: Step No(g) Ox(g) + NOz(g) + O(g) Step 2: NOz(g) O(g) No(g) 0xg Overall: Ni Write the rate law for

2. Reaction energy profiles for one-, two-, and three-step reactions... | Download Scientific Diagram

Draw an energy diagram for a two-step reaction, A to B to C, where the relative energy of these compounds is C less than A less than B, and the conversion of