Pharmaceutical toxicology: Designing studies to reduce animal use, while maximizing human translation

Immunoaffinity nanogold coupled with direct analysis in real time (DART) mass spectrometry for analytical toxicology - Analytical Methods (RSC Publishing)
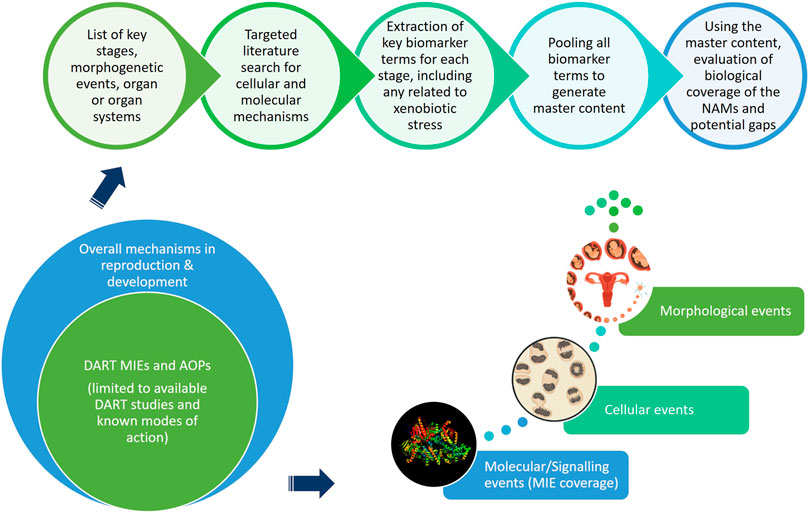
Developmental and Reproductive Toxicology (DART) - HESI - Health and Environmental Sciences Institute

DART - "Developmental And Reproductive Toxicology (database / base de données)" by AcronymsAndSlang.com

Developmental And Reproductive Toxicology (DART) Studies What Are They And How Do They Fit Into Your Program

Developmental and Reproductive Toxicology (DART) - HESI - Health and Environmental Sciences Institute

Characterization of bone abnormalities from micro-CT images for evaluating drug toxicity in developmental and reproductive toxicology (DART) studies | Semantic Scholar

Carcinogenicity *General Toxicology **DART **Safety Pharmacology HighMedium Low Genetic Toxicity Pharmacokinetics Pharmacology Study Group Priority Study. - ppt download

Considerations for Assessment of Reproductive and Developmental Toxicity of Oligonucleotide-Based Therapeutics | Nucleic Acid Therapeutics