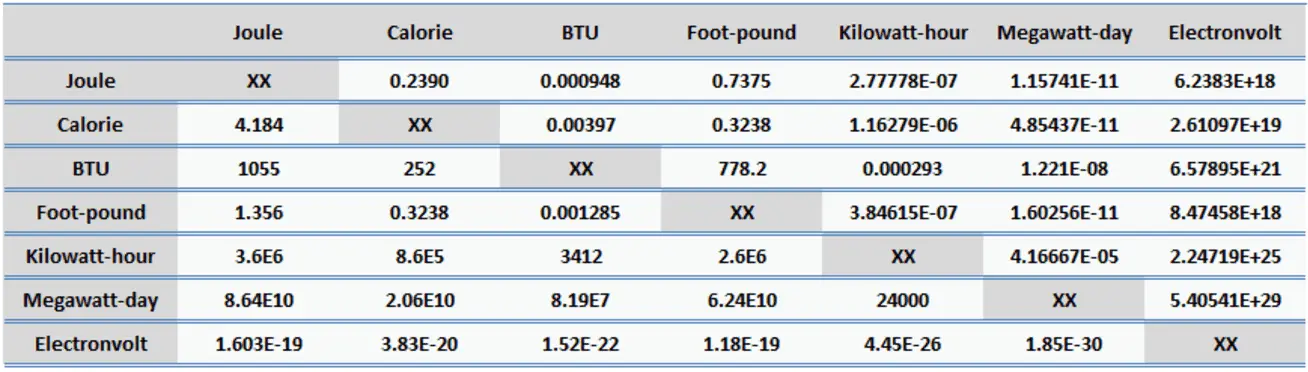
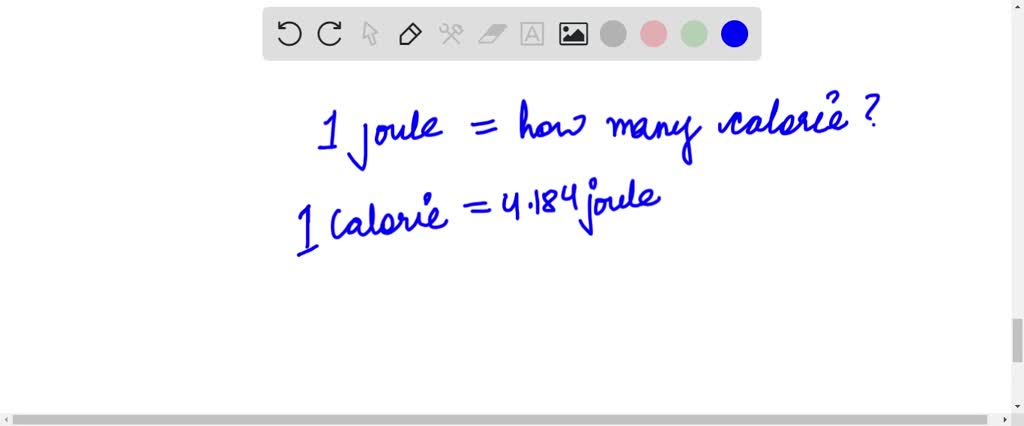
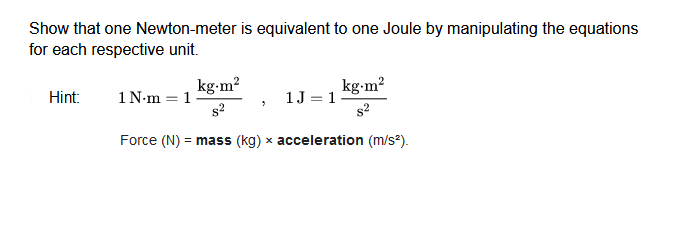Thermochemistry! AP Chapter 5. Temperature vs. Heat Temperature is the average kinetic energy of the particles in a substance. Heat is the energy that. - ppt download

1 joule of energy is to be converted into new system of units in which length is measured in 10 - YouTube
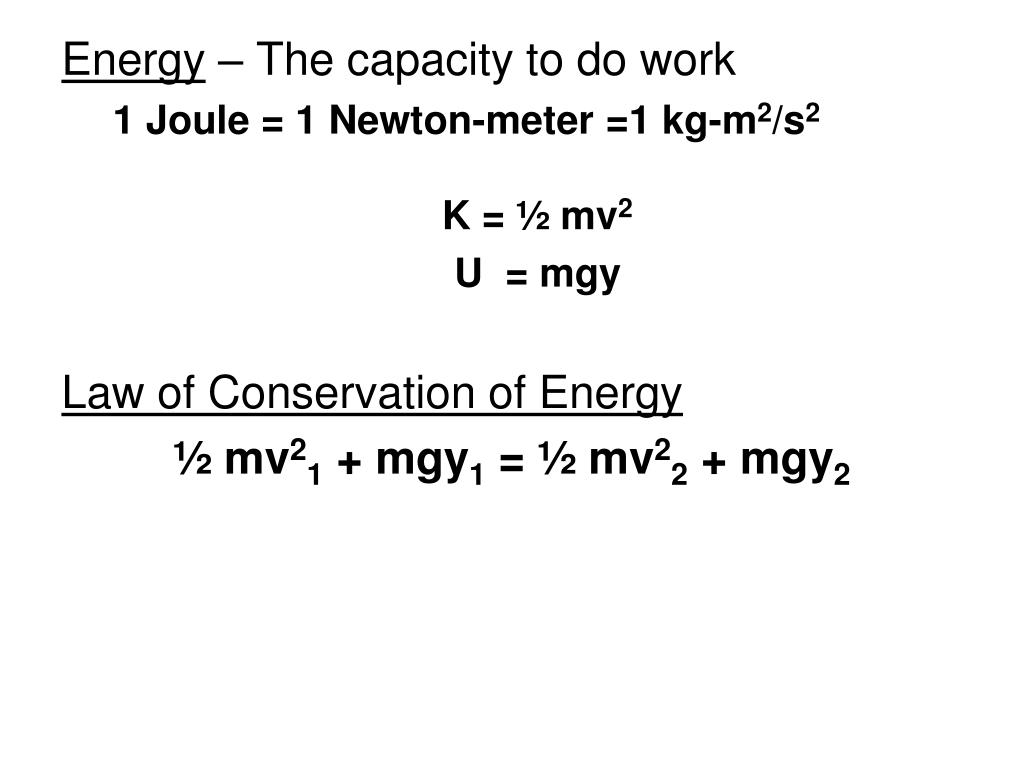
PPT - Energy – The capacity to do work 1 Joule = 1 Newton-meter =1 kg-m 2 /s 2 K = ½ mv 2 U = mgy PowerPoint Presentation - ID:6315164